...or at least those on Monday -- which is to say most of the important ones, dealing specifically with the latest results from ER and Mars Express. I've gotten another assignment from "Astronomy" to do a one-page piece on each of those subjects. (I would have loved to hang around for the remainder of the meeting to cover the Saturn and Titan meetings in particular -- but, alas I wasn't offered enough to even cover my motel expenses for that, since those meetings are spread across three more days. I do, however, intend to take careful note of the Saturn and Titan posters available on the first day.)
It's awfully late for me to give you this news -- but does anyone have any particular questions you'd like me to look into while I'm there?
Full Version: I'm Off To The Agu's Mars Sessions...
Obviously - IDD health for Opportunity 
Doug
Doug
While I didn't catch every last half-garbled word on NASA's rebroadcast of the ESA webcast of the Huygens/Mars Express briefing, I don't think I've heard a clear result of the scramble to get more radio telescopes up and listening to catch Huygen's last whisper. When was the last known signal heard, when do they think the last hearable signal reached Earth, and when do they have no-signal data confirming it was no longer transmitting? (or in terms of time after landing, etc.)
Is it possible to confirm which Cassini flyby will have Ontario Lacus radar imaged?
Roughly what is the route for Spirit in the next few months? how far down? distance/timeing to Home Plate?
Timing and targets for the next Mars Express MARSIS subsurface soundings?
Any update on the presumed "lake" on Titan or other current liquid bodies of methane?
Timing and targets for the next Mars Express MARSIS subsurface soundings?
Any update on the presumed "lake" on Titan or other current liquid bodies of methane?
Bruce,
I would be interested to hear how the MARSIS and SHARAD instruments are going to work in tandem to enhance each others science output.
I would be interested to hear how the MARSIS and SHARAD instruments are going to work in tandem to enhance each others science output.
QUOTE (Decepticon @ Dec 4 2005, 05:44 AM)
I can tell you that one. None. We go right over Ontario during T38, which would allow for altimetry, if RADAR is given prime coverage at closest approach. VIMS also could get prime, which would allow for 1-2 km/pixel imaging of the feature. During T39, RADAR SAR will get a view near Ontario Lacus, but not on the feature. There do appear to be a number of dark patches in the south polar region that may be smaller lakes or playas..
To late to ask at the meeting but perhaps you can ask via email of Mars atmospheric scientists this question.
The water vapor content of Mars atmosphere is given as around 10 precipitable microns near the equator. However, there are frequently dense clouds over some equatorial sites that I believe make this amount untenable. One such site is Noctis Labyrinthus:
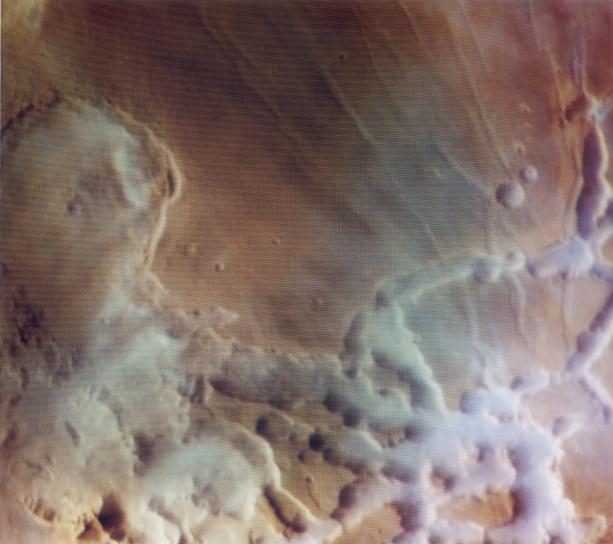

The clouds/fogs here have the appearance of precipitation carrying clouds on Earth. They do not appear for example like the thin, wispy cirrus-type clouds we've often seen on Mars such as above the MER landing sites.
The ones above Noctis look like the classical cumulus clouds that hold rain or snow on Earth.
Therefore have measurements of these clouds/fogs above Noctis been made to determine their water content?
- Bob Clark
The water vapor content of Mars atmosphere is given as around 10 precipitable microns near the equator. However, there are frequently dense clouds over some equatorial sites that I believe make this amount untenable. One such site is Noctis Labyrinthus:


The clouds/fogs here have the appearance of precipitation carrying clouds on Earth. They do not appear for example like the thin, wispy cirrus-type clouds we've often seen on Mars such as above the MER landing sites.
The ones above Noctis look like the classical cumulus clouds that hold rain or snow on Earth.
Therefore have measurements of these clouds/fogs above Noctis been made to determine their water content?
- Bob Clark
Those images have had strong contrast enhancement, but the Noctis Labrynthus clouds Viking imaged are fairly opaque. I do not see resemblence to cumulus, there's no relief on the cloud's and they are not sharp edged. Rather, they appear to be unusually dense ice-fog. Note that Labrynthus Noctis is at such high altitude, the valley floors are at lower pressure than the triple point of water and liquid is not stable at any time.
I've been frustrated that the only version of these images is the NASA press release with terrible uncorrected color shading caused by bad early-mission orbiter camera calibration files. They were never reprocessed into high quality images.
I've been frustrated that the only version of these images is the NASA press release with terrible uncorrected color shading caused by bad early-mission orbiter camera calibration files. They were never reprocessed into high quality images.
QUOTE (edstrick @ Dec 10 2005, 07:29 PM)
Those images have had strong contrast enhancement, but the Noctis Labrynthus clouds Viking imaged are fairly opaque. I do not see resemblence to cumulus, there's no relief on the cloud's and they are not sharp edged. Rather, they appear to be unusually dense ice-fog. Note that Labrynthus Noctis is at such high altitude, the valley floors are at lower pressure than the triple point of water and liquid is not stable at any time.
I've been frustrated that the only version of these images is the NASA press release with terrible uncorrected color shading caused by bad early-mission orbiter camera calibration files. They were never reprocessed into high quality images.
I've been frustrated that the only version of these images is the NASA press release with terrible uncorrected color shading caused by bad early-mission orbiter camera calibration files. They were never reprocessed into high quality images.
I couldn't find direct MOLA measurements of Noctis but I found after a google search an article that gives elevations of Noctis in profile:
Mars: Northern hemisphere slopes and slope distributions.
GEOPHYSICAL RESEARCH LETTERS, VOL. 25, NO. 24, PAGES 4413-4416, DECEMBER 15, 1998.
http://ltpwww.gsfc.nasa.gov/tharsis/grl98_slopes.pdf
Click to view attachment
You see in Fig. 3, p. 3 that the deepest portions of Noctis extend down 2 km below, perhaps up to 3 km below, the Martian 0 elevation datum. This is deeper than the Viking landing sites which showed atmospheric pressures that never fell below the 6.1 mbar triple point for water, and sometimes reached to 10 mbars.
Since these clouds are described as lying in the low lying regions of this canyon system, I take it they extend over these lowest elevation portions as well.
The primary characteristic of precipitation bearing clouds is their denseness and thickness. They don't have to be separated into individual bulbous shaped clouds. For instance the sky over an area can be overcast and the area will generally be covered by an unbroken blanket of such clouds.
Such clouds can contain liquid water or ice. You can't know which without knowing their temperature. The clouds/fogs over Noctis appear to extend down to the surface. Since this is a near equatorial site, surface temperatures can exceed 20 C, 70 F during daytime.
- Bob
http://ltpwww.gsfc.nasa.gov/tharsis/Mars_t...aphy_from_MOLA/
I stand corrected on the topography. This is in the eastern part of the Labrynthus where the large open end of the Valles Marineris is below datum. The larger image (of the labrynthus) is oriented with south up, and the densest appearing clouds (on the right) are over terrain near the zero datum and a bit higher.
Note that because of the low atmosphere pressure, air temperatures are strongly decoupled from surface temperatures. Afternoon atmosphere temperatures a few meters above the surface are something like a few tens of degrees cooler than the surface. Note that that 20 deg C surface is the Martian equivalent of the "Sidewalk you can fry an egg on" and corresponds to dark material in full early afternoon sun.
These images were taken during the morning with oblique illumination providing good relief information, and surface temperatures will be much colder than the maximum.
Also, my impression of the images is that the cloud's optical depth is near one or a bit higher, max of maybe 2 in the densest regions. I'd need the original data well processed to be sure, but my impression is the surface of the valley bottoms is visible in most areas, though at low contrast.
I stand corrected on the topography. This is in the eastern part of the Labrynthus where the large open end of the Valles Marineris is below datum. The larger image (of the labrynthus) is oriented with south up, and the densest appearing clouds (on the right) are over terrain near the zero datum and a bit higher.
Note that because of the low atmosphere pressure, air temperatures are strongly decoupled from surface temperatures. Afternoon atmosphere temperatures a few meters above the surface are something like a few tens of degrees cooler than the surface. Note that that 20 deg C surface is the Martian equivalent of the "Sidewalk you can fry an egg on" and corresponds to dark material in full early afternoon sun.
These images were taken during the morning with oblique illumination providing good relief information, and surface temperatures will be much colder than the maximum.
Also, my impression of the images is that the cloud's optical depth is near one or a bit higher, max of maybe 2 in the densest regions. I'd need the original data well processed to be sure, but my impression is the surface of the valley bottoms is visible in most areas, though at low contrast.
QUOTE (edstrick @ Dec 12 2005, 08:58 AM)
http://ltpwww.gsfc.nasa.gov/tharsis/Mars_t...aphy_from_MOLA/
I stand corrected on the topography. This is in the eastern part of the Labrynthus where the large open end of the Valles Marineris is below datum. The larger image (of the labrynthus) is oriented with south up, and the densest appearing clouds (on the right) are over terrain near the zero datum and a bit higher.
Note that because of the low atmosphere pressure, air temperatures are strongly decoupled from surface temperatures. Afternoon atmosphere temperatures a few meters above the surface are something like a few tens of degrees cooler than the surface. Note that that 20 deg C surface is the Martian equivalent of the "Sidewalk you can fry an egg on" and corresponds to dark material in full early afternoon sun.
These images were taken during the morning with oblique illumination providing good relief information, and surface temperatures will be much colder than the maximum.
Also, my impression of the images is that the cloud's optical depth is near one or a bit higher, max of maybe 2 in the densest regions. I'd need the original data well processed to be sure, but my impression is the surface of the valley bottoms is visible in most areas, though at low contrast.
I stand corrected on the topography. This is in the eastern part of the Labrynthus where the large open end of the Valles Marineris is below datum. The larger image (of the labrynthus) is oriented with south up, and the densest appearing clouds (on the right) are over terrain near the zero datum and a bit higher.
Note that because of the low atmosphere pressure, air temperatures are strongly decoupled from surface temperatures. Afternoon atmosphere temperatures a few meters above the surface are something like a few tens of degrees cooler than the surface. Note that that 20 deg C surface is the Martian equivalent of the "Sidewalk you can fry an egg on" and corresponds to dark material in full early afternoon sun.
These images were taken during the morning with oblique illumination providing good relief information, and surface temperatures will be much colder than the maximum.
Also, my impression of the images is that the cloud's optical depth is near one or a bit higher, max of maybe 2 in the densest regions. I'd need the original data well processed to be sure, but my impression is the surface of the valley bottoms is visible in most areas, though at low contrast.
Actually for salt containing brines as for example Don Juan Pond in Antarctica, *anywhere* on Mars would have sufficient surface pressure to maintain the brine in liquid form. This fact is discussed here:
On the possibility of liquid water on present-day Mars.
Haberle, Mckay, Schaeffer, Cabrol, Grin, Zent, and Quinn.
Journal of Geophysical Research, no. E10, p. 23,317-23,326, Oct. 25, 2001
It mentions that not only is the freezing point depressed by the addition of salts but so also is the equilibrium vapor pressure. That is, the required pressure for the water to stay liquid will be reduced:
"We now examine the effect of dissolved salts on the potential for melting. Pure water is unlikely on Mars since salts are believed to be a significant component of the Martian soil [Clark and Van Hart, 1981]. The presence of salts will lower the melting point and reduce the equilibrium vapor pressure of the solution. An example of the effect of a NaCl brine on the potential for melting is shown in Figure 7. In this example, the eutectic point is 251 K and the equilibrium vapor pressure of the solution at that temperature is 1.23 mbar. Clearly, the presence of salts greatly expands the regions where melting could occur and increases the total time such conditions might exist. In this particular example, virtually the entire planet (except the polar regions) experiences conditions favorable for melting at some point during the year, including the Tharsis plateau.
...
"For the NaCl brine mentioned above, boiling would not occur at its eutectic anywhere on Mars since the surface pressure never falls below 2.60 mbar anywhere on the planet during the year."
On the possibility of liquid water on present-day Mars, p. 23,321-23,322.
Bob Clark
Unfortunately, I have nothing specific to report on Mars Express' weather observations (except that it has seen signs of water ice clouds as high as 90 km, and that it confirmed the expected anti-correlation between ozone and water vapor in the air -- the first speaker failed to show up on time, and I was planning to light out at that point anyway.
But there is one interesting and perhaps relevant point which I'm now free to mention, because it won't be used in "Astronomy's" version of my MEx article: OMEGA has so far failed to find any detectable trace of iron hydroxides on the surface, even as very small deposits like the clays. There are probably some such deposits too small for it to see -- after all, MER-A has confirmed that most of the West Spur rocks in the Columbia Hills are the "Clovis" type, which is rich in goethite -- but this does seem to imply that very little of Mars' surface has been exposed to long-lasting amounts of liquid water at any time in the last few billion years. One would think that frequent wetting of the surface soil by mists in Valles Marineris would produce deposits of such hydroxides.
But there is one interesting and perhaps relevant point which I'm now free to mention, because it won't be used in "Astronomy's" version of my MEx article: OMEGA has so far failed to find any detectable trace of iron hydroxides on the surface, even as very small deposits like the clays. There are probably some such deposits too small for it to see -- after all, MER-A has confirmed that most of the West Spur rocks in the Columbia Hills are the "Clovis" type, which is rich in goethite -- but this does seem to imply that very little of Mars' surface has been exposed to long-lasting amounts of liquid water at any time in the last few billion years. One would think that frequent wetting of the surface soil by mists in Valles Marineris would produce deposits of such hydroxides.
Bruce,
I would be interested to hear how the MARSIS and SHARAD instruments are going to work in tandem to enhance each others science output.
I would be interested to hear how the MARSIS and SHARAD instruments are going to work in tandem to enhance each others science output.
There's a good new LPSC abstract on precisely that at http://www.lpi.usra.edu/meetings/lpsc2006/pdf/1450.pdf , where the polar layered deposits are concerned. I also have a document in my records somewhere on the specific nature of SHARAD's scientific purposes, once I dig it up.
That LPSC abstract also points out that, when you look more carefully at the one cross-section MARSIS has obtained so far of the northern polar layered deposits, you do see the same sort of internal layering that is far more clearly visible in the deeper southern deposits -- something that was not mentioned at all at the December AGU presentation.
This is a "lo-fi" version of our main content. To view the full version with more information, formatting and images, please click here.
